What Is the Phase Change From a Solid to a Gas Called?
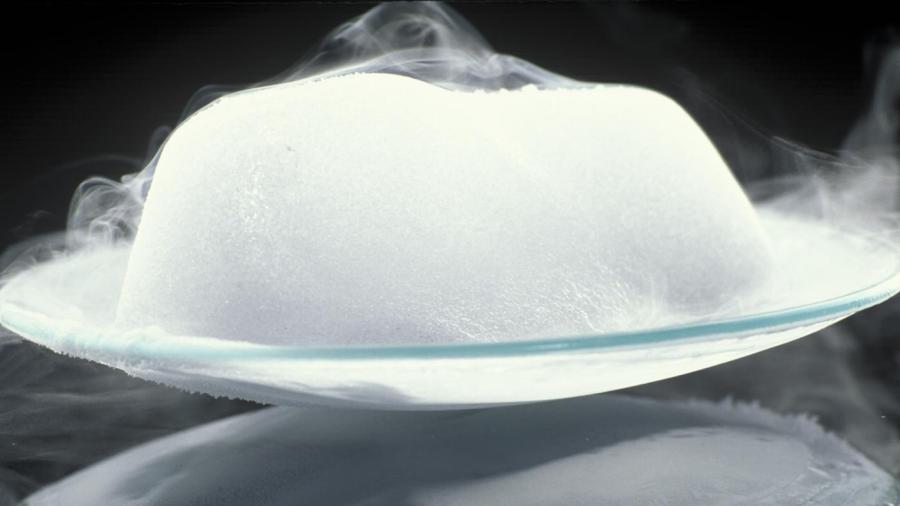
Sublimation is the change in phase from a solid to a gas. It is important to note that no liquid phase is present in sublimation.
Sublimation occurs at temperatures below a substance’s ‘triple point’. The triple point is an instant in which all three phases of matter exist in equilibrium. Triple points are represented on phase diagrams. In sublimation the solid substance is heated to a point at which it turns to gas without becoming a liquid first.
Sublimation is used to purify substances in chemical processes. Usually the practice begins by placing a solid in a vessel and then heating it within a vacuum. The pressure and heat volatilize the solid, converting the compound into a gas. The impurities remain a solid in the vessel.
Sublimation is applied to the functions of dyeing and freezing. In dyes, the ink is vaporized by a heater and then cooled on the paper or fabric. As a result the color transfer is highly-defined and more easily controlled, making more intricate dying possible.
In freezers a combination of sub-zero temperatures and dry, arid air prevent frost from forming on the inside. This is managed by an internal thermostat and fan. Iodine crystals and carbon dioxide are common examples of sublimation as they sublimate at room temperature.





