What Factors Affect the Permeability of a Cell Membrane?
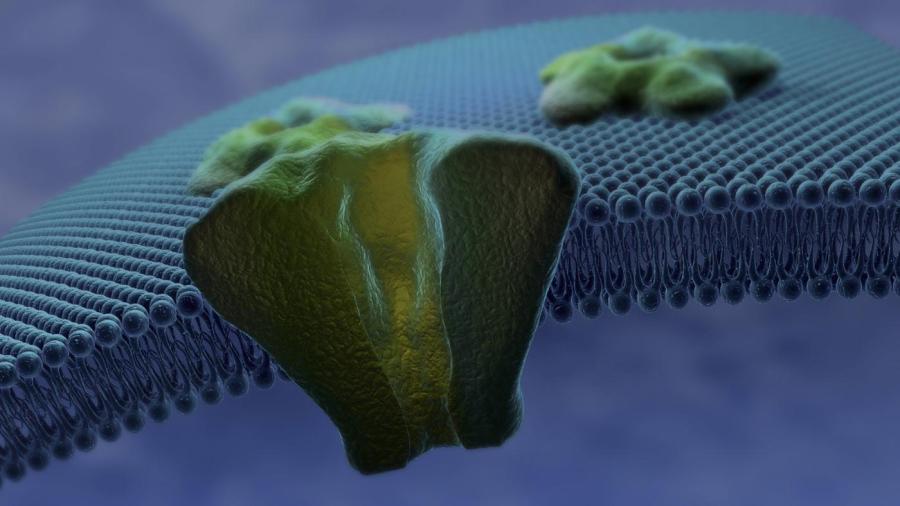
The permeability of a cell membrane is affected by the polarity, electric charge and molar mass of the molecules that diffuse through it. The phosolipid layers that make up the cell membrane also affect its permeability.
A cell membrane consists of two phosolipid layers. Each layer has an electrically charged and hydrophilic head, while the tail is uncharged and hydrophobic. The electrically charged heads of these layers face toward the water. The uncharged tails face each other. This makes it easier for small, neutrally-charged molecules to pass through the cell membrane as opposed to charged and larger molecules. The phosolipid layers also prevent non-lipid soluble substances from passing through the cell membrane.
Cell membranes are selectively permeable, allowing some substances to pass through while restricting the passage of others, says Physiology Web. This is essential for providing a cell with nutrients, eliminating waste and preventing unwanted molecules from entering a cell. The double phospholipid layers of a cell membrane include polar heads and non-polar tails, says WiseGeek. Cell membranes are very permeable to non-polar molecules, such as oxygen, nitrogen, carbon dioxide and steroids, says Physiology Web. Conversely, membranes are less permeable to small polar molecules, such as water, glycerol, urea and ethanol, and highly impermeable to large polar molecules, such as glucose and sucrose.
Electric charge also plays an important role in membrane permeability, says Physiology Web. Charged particles, or ions, cannot penetrate a cell membrane. These charged particles, such as sodium, potassium, calcium, hydrogen and chlorine ions, require specialized transport proteins to carry them across the membrane. Transport proteins also carry molecules like glucose, water and ethanol across membranes. This means that the cell has more control over how many of these molecules pass through, and how often.





